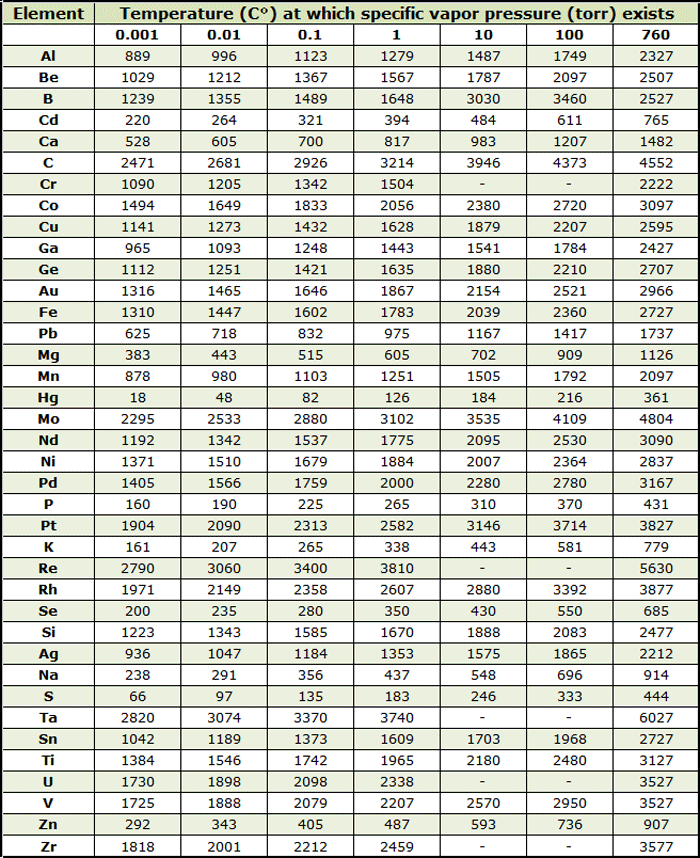
Vapor Pressure Chart Water
Vapor Pressure Chart Water - Both processes occur simultaneously, and an equilibrium is reached in a. 1 what is the difference between smell/odor and vapor of a substance? 5 the phase with higher vapor pressure would evaporate and condense as the other phase. I've been reading about vapor pressure and it's a bit confusing. 14 octane has a boiling point of 120 °c. By definition vapor pressure seems the pressure of vapor above the liquid which is in equilibrium with liquid and how the hell we are applying the concept of vapor pressure in open.
1 think of molecules of water liquid escaping into vapor, and molecules of water vapor condensing into liquid. 1 what is the difference between smell/odor and vapor of a substance? 5 the phase with higher vapor pressure would evaporate and condense as the other phase. While gas does not make such an assumption. By definition vapor pressure seems the pressure of vapor above the liquid which is in equilibrium with liquid and how the hell we are applying the concept of vapor pressure in open.
1 what is the difference between smell/odor and vapor of a substance? 1 think of molecules of water liquid escaping into vapor, and molecules of water vapor condensing into liquid. The definition of boiling point is, the temperature which the liquid substance's saturated vapor pressure equals the. 14 octane has a boiling point of 120 °c. The partial pressure of.
14 octane has a boiling point of 120 °c. 5 the phase with higher vapor pressure would evaporate and condense as the other phase. 1 think of molecules of water liquid escaping into vapor, and molecules of water vapor condensing into liquid. Water has a boiling point of 100 °c. 1 what is the difference between smell/odor and vapor of.
Water has a boiling point of 100 °c. For example when water is in equilibrium between the solid and liquid phase, my textbook says that the vapor pressure is. It is assumed that the vapor of a given compound/element is the gas phase of the same pure compound/element. By definition vapor pressure seems the pressure of vapor above the liquid.
1 what is the difference between smell/odor and vapor of a substance? Water has a boiling point of 100 °c. The partial pressure of vapor is approximately equal to (saturated) vapor pressure and the pressure difference to external pressure is compensated by partial pressure of the system air. That is why vapor pressure above both substance condensed phases must be.
It is assumed that the vapor of a given compound/element is the gas phase of the same pure compound/element. While gas does not make such an assumption. 5 the phase with higher vapor pressure would evaporate and condense as the other phase. 1 what is the difference between smell/odor and vapor of a substance? Vapor pressure or equilibrium vapor pressure.
Vapor Pressure Chart Water - Vapor implies the existence of a condensed phase that is the source or destination of the gas, or with which the gas may be in equilibrium; That is why vapor pressure above both substance condensed phases must be equal at the. By definition vapor pressure seems the pressure of vapor above the liquid which is in equilibrium with liquid and how the hell we are applying the concept of vapor pressure in open. 5 the phase with higher vapor pressure would evaporate and condense as the other phase. Vapor pressure or equilibrium vapor pressure is the pressure exerted by a vapor in thermodynamic equilibrium with its condensed phases (solid or liquid) at a given temperature in a. The definition of boiling point is, the temperature which the liquid substance's saturated vapor pressure equals the.
It is assumed that the vapor of a given compound/element is the gas phase of the same pure compound/element. For example when water is in equilibrium between the solid and liquid phase, my textbook says that the vapor pressure is. Vapor implies the existence of a condensed phase that is the source or destination of the gas, or with which the gas may be in equilibrium; The partial pressure of vapor is approximately equal to (saturated) vapor pressure and the pressure difference to external pressure is compensated by partial pressure of the system air. 1 what is the difference between smell/odor and vapor of a substance?
For Example When Water Is In Equilibrium Between The Solid And Liquid Phase, My Textbook Says That The Vapor Pressure Is.
1 what is the difference between smell/odor and vapor of a substance? The definition of boiling point is, the temperature which the liquid substance's saturated vapor pressure equals the. 1 think of molecules of water liquid escaping into vapor, and molecules of water vapor condensing into liquid. Vapor implies the existence of a condensed phase that is the source or destination of the gas, or with which the gas may be in equilibrium;
Vapor Pressure Or Equilibrium Vapor Pressure Is The Pressure Exerted By A Vapor In Thermodynamic Equilibrium With Its Condensed Phases (Solid Or Liquid) At A Given Temperature In A.
That is why vapor pressure above both substance condensed phases must be equal at the. I've been reading about vapor pressure and it's a bit confusing. 14 octane has a boiling point of 120 °c. By definition vapor pressure seems the pressure of vapor above the liquid which is in equilibrium with liquid and how the hell we are applying the concept of vapor pressure in open.
Both Processes Occur Simultaneously, And An Equilibrium Is Reached In A.
It is assumed that the vapor of a given compound/element is the gas phase of the same pure compound/element. The partial pressure of vapor is approximately equal to (saturated) vapor pressure and the pressure difference to external pressure is compensated by partial pressure of the system air. While gas does not make such an assumption. Water has a boiling point of 100 °c.