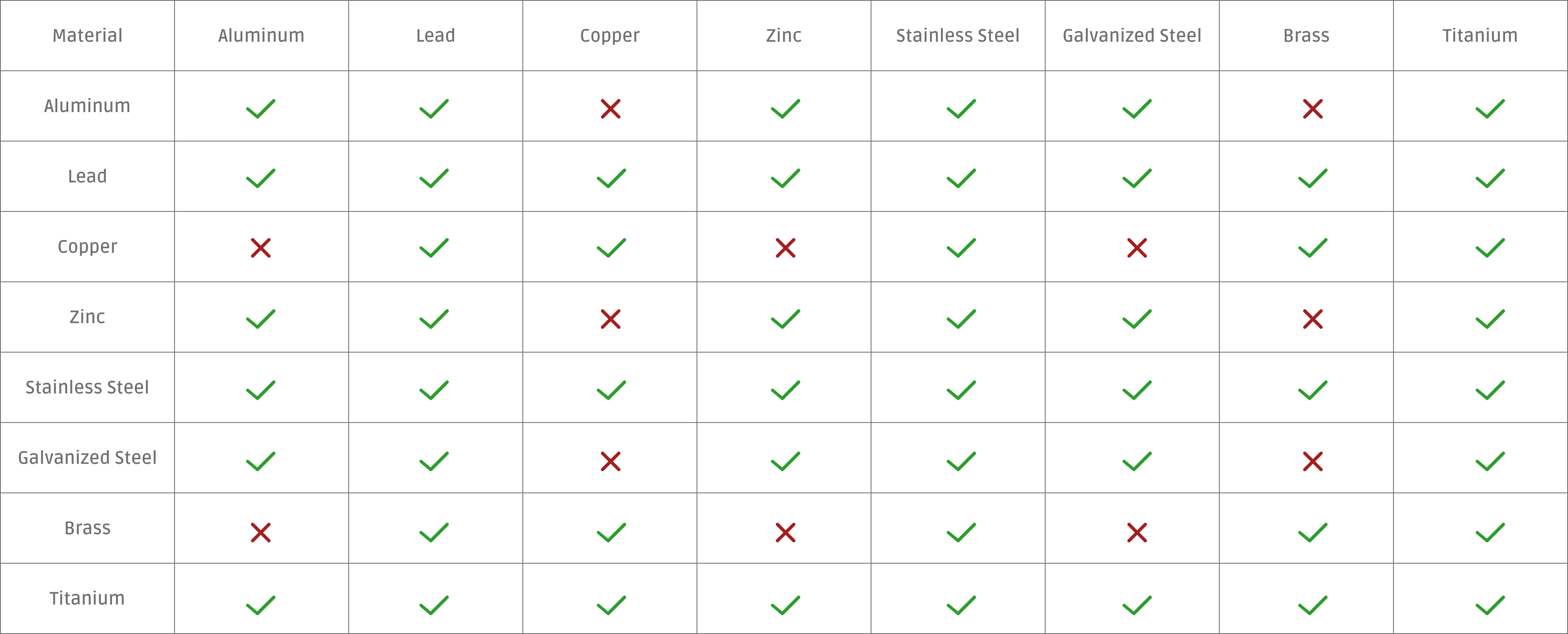
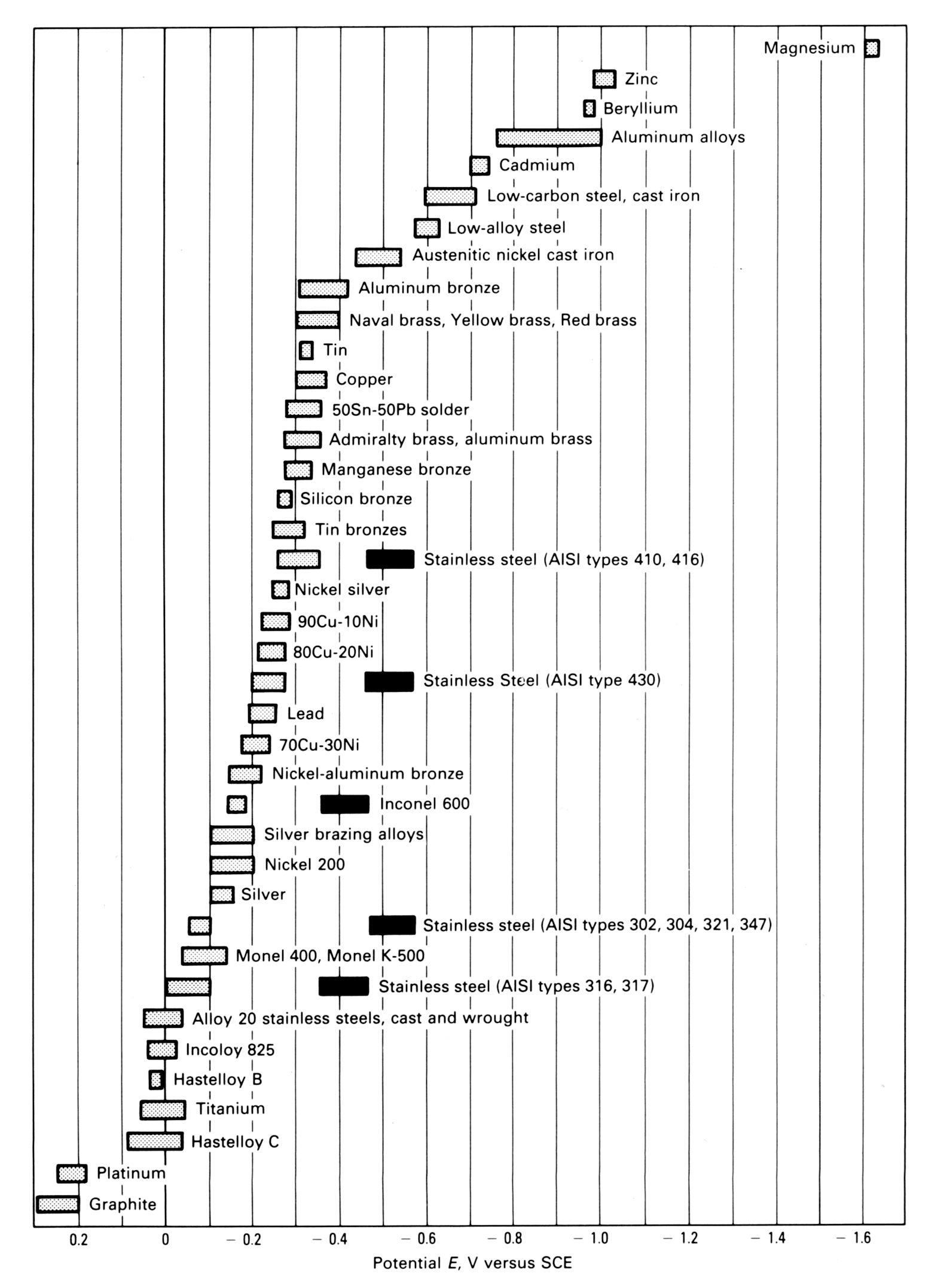
Galvanic Scale Chart
Galvanic Scale Chart - Producing or caused by an electric current. It’s one of the most common causes of. Unlike a direct redox reaction, where chemical energy is primarily released as heat, a galvanic cell converts chemical energy into electrical energy. Galvanic cell, electrolytic cell, and dry cell electrochemical cells are devices that either produce electrical energy from a chemical reaction or use electrical energy to cause a chemical reaction. This page explains the roles of the anode, cathode, and the salt bridge. Pertaining to or produced by galvanism;
A galvanic or voltaic cell is an electrochemical cell that converts chemical energy into electrical energy. However, this conversion is not 100% efficient, and. Producing or caused by an electric current. Galvanic corrosion is an electrochemical process that eats away at one metal when two different metals are in contact with each other in the presence of moisture. This page explains the roles of the anode, cathode, and the salt bridge.
This page explains the roles of the anode, cathode, and the salt bridge. However, this conversion is not 100% efficient, and. The meaning of galvanic is of, relating to, or producing a direct current of electricity. See examples of galvanic used in a sentence. A galvanic or voltaic cell is an electrochemical cell that converts chemical energy into electrical energy.
This page explains the roles of the anode, cathode, and the salt bridge. However, this conversion is not 100% efficient, and. Galvanic corrosion is an electrochemical process that eats away at one metal when two different metals are in contact with each other in the presence of moisture. How to use galvanic in a sentence. A galvanic or voltaic cell.
Galvanic cell, electrolytic cell, and dry cell electrochemical cells are devices that either produce electrical energy from a chemical reaction or use electrical energy to cause a chemical reaction. Producing or caused by an electric current. However, this conversion is not 100% efficient, and. This page explains the roles of the anode, cathode, and the salt bridge. Galvanic cells operate.
Galvanic corrosion is an electrochemical process that eats away at one metal when two different metals are in contact with each other in the presence of moisture. Unlike a direct redox reaction, where chemical energy is primarily released as heat, a galvanic cell converts chemical energy into electrical energy. This page explains the roles of the anode, cathode, and the.
Galvanic cell, electrolytic cell, and dry cell electrochemical cells are devices that either produce electrical energy from a chemical reaction or use electrical energy to cause a chemical reaction. The meaning of galvanic is of, relating to, or producing a direct current of electricity. However, this conversion is not 100% efficient, and. A galvanic cell or voltaic cell, named after.
Galvanic Scale Chart - It achieves this by harnessing the energy produced by the redox reactions that. A galvanic or voltaic cell is an electrochemical cell that converts chemical energy into electrical energy. A galvanic cell or voltaic cell, named after the scientists luigi galvani and alessandro volta, respectively, is an electrochemical cell in which an electric current is generated from spontaneous. Galvanic corrosion is an electrochemical process that eats away at one metal when two different metals are in contact with each other in the presence of moisture. Pertaining to or produced by galvanism; It’s one of the most common causes of.
Galvanic cell, electrolytic cell, and dry cell electrochemical cells are devices that either produce electrical energy from a chemical reaction or use electrical energy to cause a chemical reaction. The meaning of galvanic is of, relating to, or producing a direct current of electricity. Producing or caused by an electric current. See examples of galvanic used in a sentence. A galvanic or voltaic cell is an electrochemical cell that converts chemical energy into electrical energy.
Producing Or Caused By An Electric Current.
See examples of galvanic used in a sentence. This page explains the roles of the anode, cathode, and the salt bridge. However, this conversion is not 100% efficient, and. Galvanic cells operate because oxidation and reduction occur in separate locations, forcing electrons through an external circuit.
A Galvanic Cell Or Voltaic Cell, Named After The Scientists Luigi Galvani And Alessandro Volta, Respectively, Is An Electrochemical Cell In Which An Electric Current Is Generated From Spontaneous.
It achieves this by harnessing the energy produced by the redox reactions that. Galvanic cell, electrolytic cell, and dry cell electrochemical cells are devices that either produce electrical energy from a chemical reaction or use electrical energy to cause a chemical reaction. Pertaining to or produced by galvanism; Unlike a direct redox reaction, where chemical energy is primarily released as heat, a galvanic cell converts chemical energy into electrical energy.
Galvanic Corrosion Is An Electrochemical Process That Eats Away At One Metal When Two Different Metals Are In Contact With Each Other In The Presence Of Moisture.
The meaning of galvanic is of, relating to, or producing a direct current of electricity. How to use galvanic in a sentence. It’s one of the most common causes of. A galvanic or voltaic cell is an electrochemical cell that converts chemical energy into electrical energy.
![Galvanic Corrosion [with Chart] EngineerExcel](https://i2.wp.com/engineerexcel.com/wp-content/uploads/2023/03/galvanic-corrosion-chart.png)