Galvanic Corrosion Chart
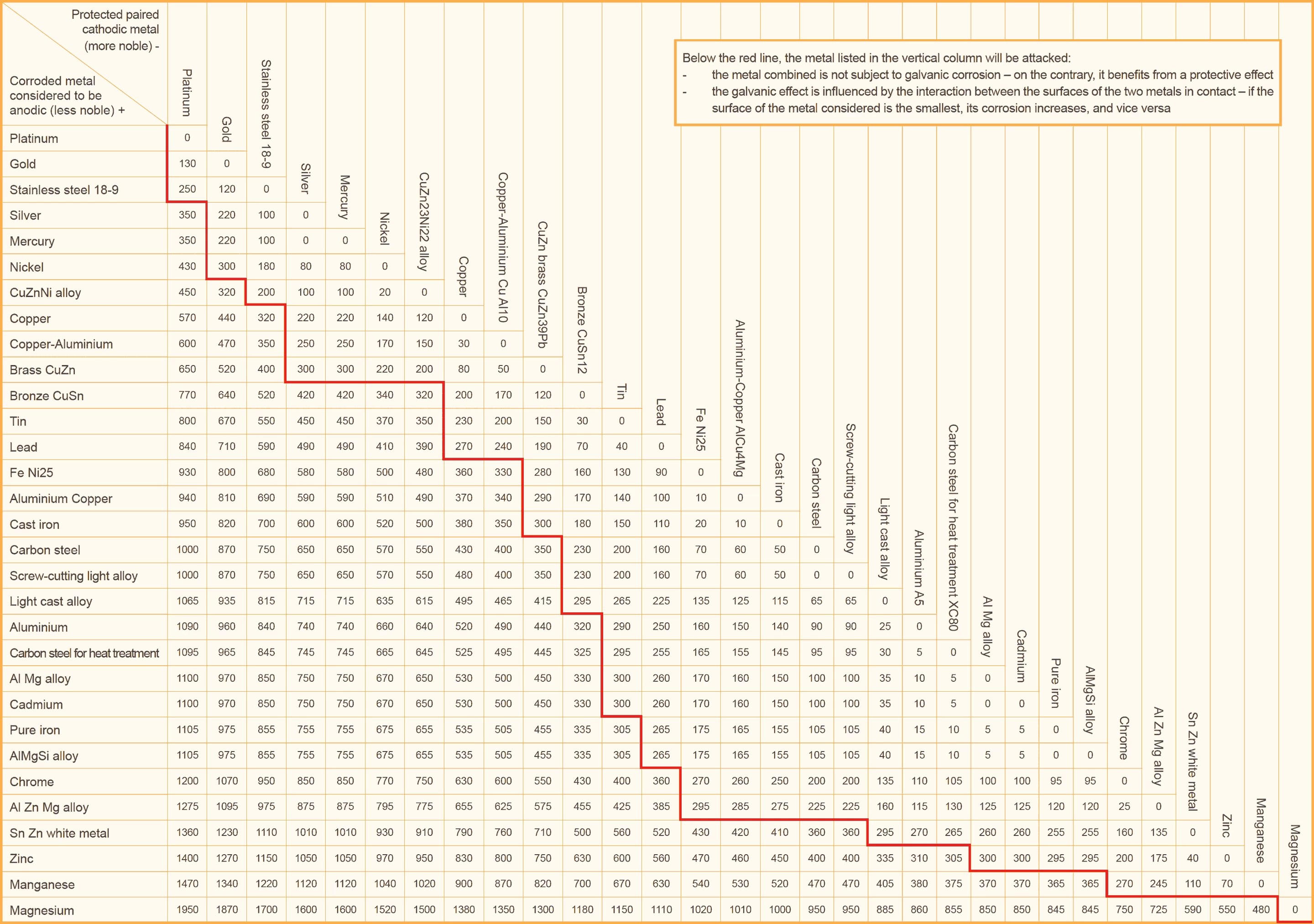
Galvanic Corrosion Chart - This chart is designed to assist in broadly assessing the risk of galvanic corrosion associated with a given metal coming into contact with another metal. An anode, cathode, electrolyte, and return path. Fastener material selection based on the galvanic series of metals table developed using information supplied by aisi committee of stainless steel producers. To assess the risk of a metallic connection, the first step is to locate both materials on the galvanic series chart specific to the intended environment. Galvanic corrosion occurs at higher rates when dissimilar metals are in contact. For example, connecting aluminum and.
To assess the risk of a metallic connection, the first step is to locate both materials on the galvanic series chart specific to the intended environment. A typical rule of thumb is that voltage differences of 0.2 volts. Galvanic corrosion occurs at higher rates when dissimilar metals are in contact. Galvanic potential the table below reports the corrosion potentials or galvanic series of metals in flowing sea water at ambient temperature. This chart is designed to assist in broadly assessing the risk of galvanic corrosion associated with a given metal coming into contact with another metal.
There are three conditions that must exist for. This chart is designed to assist in broadly assessing the risk of galvanic corrosion associated with a given metal coming into contact with another metal. Fastener material selection based on the galvanic series of metals table developed using information supplied by aisi committee of stainless steel producers. For example, connecting aluminum and..
To assess the risk of a metallic connection, the first step is to locate both materials on the galvanic series chart specific to the intended environment. These charts show which commonly used metals are compatible and which will result in galvanic corrosion when in contact. Fastener material selection based on the galvanic series of metals table developed using information supplied.
These charts show which commonly used metals are compatible and which will result in galvanic corrosion when in contact. To assess the risk of a metallic connection, the first step is to locate both materials on the galvanic series chart specific to the intended environment. An anode, cathode, electrolyte, and return path. Explore our detailed anodic index table to select.
Galvanic corrosion (some times called dissimilar metal corrosion) is the process by which the materials in contact with each other oxidizes or corrodes. An anode, cathode, electrolyte, and return path. A typical rule of thumb is that voltage differences of 0.2 volts. Explore our detailed anodic index table to select appropriate metals and protect your structural components. The galvanic corrosion.
These charts show which commonly used metals are compatible and which will result in galvanic corrosion when in contact. For galvanic corrosion to occur, four elements are necessary: A typical rule of thumb is that voltage differences of 0.2 volts. To assess the risk of a metallic connection, the first step is to locate both materials on the galvanic series.
Galvanic Corrosion Chart - This chart is designed to assist in broadly assessing the risk of galvanic corrosion associated with a given metal coming into contact with another metal. Galvanic potential the table below reports the corrosion potentials or galvanic series of metals in flowing sea water at ambient temperature. There are three conditions that must exist for. An anode, cathode, electrolyte, and return path. The galvanic series chart below shows metals and their electrochemical voltage range (relative activity in flowing sea water). Galvanic corrosion occurs at higher rates when dissimilar metals are in contact.
Fastener material selection based on the galvanic series of metals table developed using information supplied by aisi committee of stainless steel producers. Below, we give a brief overview of galvanic corrosion and provide a galvanic corrosion chart to help fabricators and machinists avoid using the wrong metal combinations. This chart is designed to assist in broadly assessing the risk of galvanic corrosion associated with a given metal coming into contact with another metal. The galvanic series chart below shows metals and their electrochemical voltage range (relative activity in flowing sea water). An anode, cathode, electrolyte, and return path.
Galvanic Potential The Table Below Reports The Corrosion Potentials Or Galvanic Series Of Metals In Flowing Sea Water At Ambient Temperature.
For example, connecting aluminum and. The galvanic series chart below shows metals and their electrochemical voltage range (relative activity in flowing sea water). These charts show which commonly used metals are compatible and which will result in galvanic corrosion when in contact. Galvanic corrosion (some times called dissimilar metal corrosion) is the process by which the materials in contact with each other oxidizes or corrodes.
Explore Our Detailed Anodic Index Table To Select Appropriate Metals And Protect Your Structural Components.
There are three conditions that must exist for. Galvanic corrosion occurs at higher rates when dissimilar metals are in contact. An anode, cathode, electrolyte, and return path. For galvanic corrosion to occur, four elements are necessary:
This Chart Is Designed To Assist In Broadly Assessing The Risk Of Galvanic Corrosion Associated With A Given Metal Coming Into Contact With Another Metal.
The galvanic corrosion process is a transfer of electrons between two electrodes through an. Fastener material selection based on the galvanic series of metals table developed using information supplied by aisi committee of stainless steel producers. Below, we give a brief overview of galvanic corrosion and provide a galvanic corrosion chart to help fabricators and machinists avoid using the wrong metal combinations. To assess the risk of a metallic connection, the first step is to locate both materials on the galvanic series chart specific to the intended environment.


![Galvanic Corrosion [with Chart] EngineerExcel](https://i2.wp.com/engineerexcel.com/wp-content/uploads/2023/03/galvanic-compatibility.webp)